
If you’re struggling to remember when you last emptied your bowels, and you vaguely recall having to strain before you managed to pass out something (which was likely lumpy, dry, and hard), you may be dealing with constipation.
While constipation isn’t typically life-threatening, it’s undeniably uncomfortable.
Averaging just one or two bowel movements a week can make you feel bloated, crampy, and sluggish. All that straining may also cause several painful complications, including anal fissures [1] and rectal prolapse [2].
Fortunately, resistant maltodextrin, a type of prebiotic fiber, can help “regularize” the frequency of your bowel movements and make those stools easier to pass. Continue reading to find out how.
Constipation is a condition in which an individual has uncomfortable or infrequent bowel movements. Symptoms include:
-Having fewer than three bowel movements per week (note: most gastroenterologists consider anywhere between 3 to 21 movements a week “normal”)
-Straining or lumpy, hard stools for more than 25% of defecation attempts
-Experiencing a feeling of incomplete elimination after a bowel movement
The Bristol stool chart [4] is a useful tool to evaluate your stools’ appearance and determine whether you’re constipated.
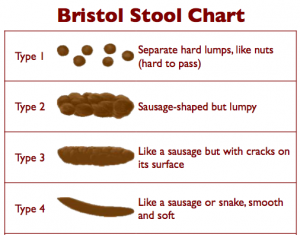
Under normal circumstances, if you’re not constipated, your stools should resemble types 3 and 4 — with a sausage-like shape, either with cracks on the surface or a smooth and soft surface.
On the flip side, if you’re constipated, your stools will look like types 1 and 2: Hard, dry, and lumpy [4].
In general, constipation can occur when the movement and contractions of the digestive tract are too slow, resulting in more water absorption from the stools by the large intestine.
When the stools reach the colon, they become hard and dry, requiring greater effort to pass out or may even result in blockage. In severe cases, medical interventions will be necessary to remove the stools and clear the blockage.
To understand how resistant maltodextrin, a prebiotic fiber, keeps constipation away, you'll first need to know what it is, so let's start there.
Prebiotic fiber (also known simply as "prebiotics") is an indigestible carbohydrate found in many fruits and vegetables, particularly those that contain complex carbohydrates, such as resistant maltodextrin [5].
The most immediate mechanism through which resistant maltodextrin helps prevent constipation is through its high water-drawing effect in the colon. This draws water from the surrounding membrane to the stools, which softens the stools and facilitates their movement down the large intestine and out of the digestive tract [6].
This is especially useful for people suffering from severe forms of constipation with very dry and hardened stools. The water-drawing effect of resistant maltodextrin enables it to draw water to the stools, softening them and making them easier to be passed out instead of adding bulk which may worsen constipation.
In addition to its water-drawing effect, resistant maltodextrin also undergoes a process known as fermentation when it resists digestion by the body’s enzymes and reaches the colon.
Fermentation occurs when the gut microbiota feeds on prebiotic fibers, producing an array of beneficial metabolites. One of the many metabolites is short-chain fatty acids (SCFAs) [7].
SCFAs are energy sources for cells in the surrounding tissues of the colon.
They can stimulate the large intestine and increase peristaltic contractions [8], ensuring that waste products can transit through the digestive tract and out of the body smoothly.
The remaining waste products from the fermentation process also add bulk to the stools, which makes them easier to be passed out.
Finally, while your body can't break down prebiotic fibers into simple sugars for energy, they are a food source for the good gut bacteria in your small intestine and colon.
Prebiotic fibers feed and nourish your good gut bacteria, which enhances the healthy balance of your gut microbiota.
The gut microbiota consists of different types of microorganisms, such as bacteria, viruses, and fungi [9]. The number of bacterial cells is estimated to outnumber our human cells by about 10 times.
Everyone is born with a unique set of innate (inborn, natural) gut microbiota. The composition and amount then get altered throughout the lifespan due to lifestyle, environmental, and dietary factors [10]. The use of medications such as antibiotics can also impact your gut microbiota.
What a healthy gut microbiota looks like can differ from person to person.
In general, however, scientists agree that a healthy gut maintains a balance between good gut bacteria and bad gut bacteria [11].
And prebiotic fibers help encourage a happy, healthy gut by feeding and nourishing your good gut bacteria — creating an environment where beneficial bacteria (such as Lactobacillus and Bifidobacterium) can flourish and crowd out unwanted and harmful bacteria.
Research [12] shows maintaining a healthy gut microbiome could prevent constipation.
To promote bowel irregularity, focus on taking at least 3-5 grams of prebiotic fiber daily.
 Probiotics vs Prebiotics: Do You Know the Differences?
Probiotics vs Prebiotics: Do You Know the Differences?
 Prebiotic: Gut Microbiota, Gut Health, and Beyond
Prebiotic: Gut Microbiota, Gut Health, and Beyond

- Metabolic health can be defined by several clinical markers, including blood pressure, blood sugar, and blood lipid levels.
- This healthy balance of gut microbiota and optimum gut health leads to improved overall well-being, including benefits to our metabolic health.
- Apart from reducing the consumption of sugar and fats, increasing the intake of prebiotics should also be considered to support metabolic health.
Metabolic health can be defined by several clinical markers, including blood pressure, blood sugar, and blood lipid levels. Metabolic syndrome refers to a cluster of biological risk factors that can lead to chronic conditions like diabetes, heart disease and other health problems [1]. It is estimated that more than 3 in 10 adults in the United States suffer from metabolic syndrome [2]. Good metabolic health can help lower your risk of developing such disorders.
Other than lifestyle changes such as engaging in regular exercises, improving sleep and managing stress, dietary changes also help support our metabolic health and processes. Apart from reducing the consumption of sugar and fats, increasing the intake of prebiotics (whether via food or supplements) should also be considered. Read on to understand more about prebiotics and how they help improve and support our metabolic health.
As defined by the International Scientific Association for Probiotics and Prebiotics (ISAPP), a prebiotic is defined as a substrate that is selectively utilized by host microorganisms conferring a health benefit [3].
Our gut consists of trillions of microorganisms including bacteria, viruses, fungi and protozoa species. This composition of microorganisms is termed the gut microbiota and is unique to everyone. It varies due to several factors such as environmental, lifestyle, dietary habits, and consumption of medications such as antibiotics [4]. An optimum gut microbiota consists of a healthy balance of both good and bad gut bacteria species.
Prebiotics feed and enable the proliferation of good gut bacteria, which in turn crowd out the bad ones. This healthy balance of gut microbiota and optimum gut health leads to improved overall well-being, including benefits to our metabolic health.
One aspect of metabolic health is blood cholesterol level. Studies have shown that a healthy balance of gut microbiota may lower blood cholesterol levels through several mechanisms. One mechanism is the metabolizing of cholesterol into coprostanol, which reduces cholesterol absorption [5,6].
Good gut bacteria feed on prebiotics in a process called fermentation, which produces short-chain fatty acids (SCFAs). SCFAs like butyrate and propionate may also have cholesterol-lowering effects by controlling and inhibiting liver cholesterol synthesis [7].
Prebiotics may also have a direct effect on lowering cholesterol levels. Prebiotics, many of which are dietary fibers, move to the colon largely unchanged and undigested. This helps to increase the viscosity of the digestive tract, leading to decreased absorption of cholesterol and blood cholesterol levels [8].
Blood sugar is another aspect of metabolic health. Apart from cholesterol levels, the metabolites of the fermentation process also influence sugar metabolism and homeostasis. SCFAs such as butyrate act as signaling molecules and activate receptors involved in secreting chemicals called glucagon-like peptide 1 (GLP-1). GLP-1 is crucial in blood glucose metabolism by increasing insulin release [9].
SCFAs also directly impact sugar metabolism, by improving glucose metabolism in the liver and increasing sugar uptake by the body cells [9].
For prebiotics which are dietary fibers, they also support healthy blood pressure. The use of fibers to reduce blood pressure has been studied and published in various clinical trials. A meta-analysis has shown that fiber supplementation at 11.5g per day was able to decrease blood pressure [10]. The Dietary Approaches to Stop Hypertension (commonly known as the DASH diet) also recommends a high-fiber intake as one of the many recommendations to reduce blood pressure.
The gut microbiota and SCFAs produced from the fermentation of prebiotics can also affect blood pressure. SCFAs activate receptors that play a part in blood pressure regulation. Unhealthy gut microbiota also leads to chronic low-grade inflammation which can be a cause of high blood pressure as well [11].
Metabolic health disorders are on the rise across the world. Besides lifestyle modifications such as regular physical activity, managing stress and having adequate sleep, dietary modifications also help. Prebiotics enhance the gut microbiota and optimize gut health, which in turn support metabolic health such as blood pressure, sugar and cholesterol levels as shown in various studies.
 Good Gut Health Is Linked To Lower Cholesterol Levels
Good Gut Health Is Linked To Lower Cholesterol Levels
 Gut Microbiota and Its Implications in Diabetes and Blood Sugar
Gut Microbiota and Its Implications in Diabetes and Blood Sugar
 Gut Health and Blood Pressure (Hypertension): How Are They Related?
Gut Health and Blood Pressure (Hypertension): How Are They Related?

While both probiotics and prebiotics have been widely studied and used, increased usage of prebiotics in recent times has reflected a greater understanding of their multi-faceted benefits to human health. They sound similar and some may even confuse one for the other. Read on to understand the functions of each.
Probiotics are defined by the International Scientific Association for Probiotics and Prebiotics (ISAPP) as live microorganisms that, when administered in adequate amounts, confer a health benefit on the host. [1] In essence, probiotic supplements contain good gut bacterial species, including Bifidobacterium and Lactobacillus species. [2]
On the other hand, prebiotics are defined by ISAPP as substrates that are selectively utilized by the host microorganisms conferring a health benefit. [3] To put it simply, prebiotics act as food for the good gut bacteria to provide health benefits.
Prebiotics such as resistant starches and various types of oligosaccharides resist digestion from our human body’s enzymes and travel to the colon largely unchanged.
Our gut microbiota in the colon and the probiotic supplements we take will then feed on and ferment the prebiotics. This fermentation process produces metabolites such as short-chain fatty acids (SCFAs) that confer health benefits such as providing a main source of energy for our colon cells and having anti-inflammatory properties. [4]
Watch the videos from ISAPP below to learn more.
[embedyt] https://www.youtube.com/watch?v= C6j7F-JhtmQ[/embedyt]
[embedyt] https://www.youtube.com/watch?v= bBrLqkpMlw8[/embedyt]
Videos by The International Scientific Association for Probiotics and Prebiotics

Your gut microbiome inhabits your digestive tract and helps break down food. So, it's only logical it'll affect your digestive health.
But… how would one explain the link between poor gut health and increased stress reactivity, alongside risk for various mental health conditions such as anxiety disorders and depression then [1]?
Well, as unrelated as the two concepts — i.e. gut health and mental well-being — may appear at first glance, they're interconnected. Read on to find out how.
Good gut health (specific strains and their amounts in the microbiome) can look wildly different from individual to individual [2]. In general, though, researchers define good gut health as [3]:
- A balance between symbiotic and pathogenic microbiome: The gut microbiome consists of microbes — bacteria, fungi, parasites, and viruses — that are both helpful and potentially harmful. Most are symbiotic (good), while some, in smaller numbers, are pathogenic (disease-promoting or bad). In a healthy body, good and bad microbes coexist without problems. But this delicate balance can be disrupted.
- High gut microbiome diversity: Studies consistently link having a diverse population of gut microbes with better health, physically (e.g., lower risk of metabolic syndrome) and emotionally [4, 5].
So, in that sense, gut dysbiosis — a phenomenon where your gut microbiome becomes unbalanced or loses diversity — is a sign of poor gut health [6].
So, why and how does gut dysbiosis impact your mental well-being? Most of it comes down to gut-driven inflammation’s adverse effects on the gut-brain axis.
See: gut dysbiosis could contribute to increased intestinal barrier permeability [7].
This may allow substances, like undigested food particles, to sneak past the intestinal lining into the bloodstream where they don’t belong — causing widespread inflammation that [8]:
1. Affects the hypothalamic-pituitary-adrenal (HPA) axis: The HPA axis' primary function is to regulate the stress response [9]. Inflammation may contribute to HPA axis dysregulation, which, in turn, is associated with chronic stress and various psychiatric conditions like depression and anxiety disorders [10].
2. Decreases levels of gut bacteria beneficial to mental well-being: Gut inflammation further exacerbates dysbiosis by promoting the growth of bad gut bacteria [11]. These crowd out mental-health-beneficial strains of probiotic gut bacteria, like species of Lactobacillus (e.g., L. paracasei, L. acidophilus, L. plantarum, and L. fermentum) that have been shown to help improve stress resilience and alleviate anxiety symptoms [12, 13, 14, 15, 16].
3. Lowers production of mood-regulating neurotransmitters: Gut inflammation could decrease the amount of acetate, propionate, and butyrate — short-chain fatty acids (SCFAs) — produced in the colon [17]. So how is this related to your mental well-being? SCFAs are involved in synthesizing several mood-regulating neurotransmitters, including serotonin, gamma-aminobutyric acid (GABA), and dopamine [18]. Put simply, gut dysbiosis could cause neurotransmitter imbalance in the brain, increasing stress reactivity, plus risk for depression and anxiety disorders [19, 20, 21].
Of course, there are many other mechanisms (via the gut-brain axis) through which gut health could influence mental well-being.
But the important takeaway is this: gut health and mental well-being are closely related.
Take care of your gut health, and your mental well-being will likely improve. How? By taking prebiotics or probiotics. To understand which of the two to take, let’s look at how they differ [22]:
- Probiotics: Live, beneficial microorganisms
- Prebiotics: Substrates that are selectively utilized by the host microorganisms conferring health benefits
As mentioned earlier, even amongst people with good gut health, no two individuals' gut microbiota will look the same. Everyone differs in the variety and abundance of species in the gut microbiota.
So, to reap the most health benefits from probiotics, you'd have to source for the specific organism(s) — genus, species, and strain — already present in your microbiome: an impossible task without gut microbiome testing.
Thankfully, you won't run into this concern with prebiotics. You can rest assured that they will feed the good gut bacteria already thriving in your gut, helping them flourish [23].
Better still, if you've undergone microbiome testing and are supplementing with probiotics native to your unique gut microbiota. Prebiotics could further improve the survivability rate of the probiotics in your digestive tract, boosting the chances that they successfully coax your gut health into a more balanced, diverse state [24].
How much prebiotics should you take, though?
According to the International Scientific Association for Probiotics and Prebiotics (ISAPP), anywhere between 3 to 5 grams would suffice [25].
Your gut and brain communicate through the gut-brain axis. So, when your gut health suffers, your mental well-being does, too.
To foster good gut health (plus keep stress, anxiety, and depression at bay), focus on gut microbiome balance and diversity. Ensuring 3-5g of prebiotics intake daily helps nourish and maintain a well-balanced gut microbiota.
 Prebiotic: Gut Microbiota, Gut Health, and Beyond
Prebiotic: Gut Microbiota, Gut Health, and Beyond

- Chronic Obstructive Pulmonary Disease (COPD) is estimated to be the third leading cause of death worldwide.
- Our gut and lungs communicate bi-directionally via the gut-lung axis. Gut health and microbiota are involved in respiratory diseases such as COPD through this gut-lung axis.
- A healthy and balanced gut microbiota may improve our respiratory health and reduce the risk of COPD.
- The use of prebiotics to induce positive changes to our gut microbiota and improve our gut health is one option we can consider to improve our respiratory health and reduce the risk of COPD.
Chronic Obstructive Pulmonary Disease (COPD) is a term used to describe a group of respiratory diseases that cause airflow blockages and breathing difficulties. COPD is associated with chronic inflammation of the airways and includes emphysema and chronic bronchitis [1]. The World Health Organization (WHO) estimates COPD to be the third leading cause of death worldwide, causing more than 3 million deaths in 2019 alone [2]. In the United States, an estimated 16 million people are diagnosed with COPD [3].
Recent studies have discovered that our gut and lungs communicate bi-directionally via the gut-lung axis. Gut health and microbiota are involved in respiratory diseases such as COPD through this gut-lung axis. Read on to find out more about how a healthy and balanced gut microbiota may improve our respiratory health and reduce the risk of COPD.
Our gut consists of trillions of such microorganisms, including bacteria, viruses, fungi and protozoa species. This set of gut microbiota is unique to everyone and varies due to several factors including environmental, lifestyle, dietary habits, and consumption of medications such as antibiotics [4].
This innate set of gut microbiota (the set that we are born with) is thought to be the most optimum for oneself. This optimum composition deteriorates as we age. Along with poorer modern-day diets lacking in prebiotics (food for the good gut bacteria) as well as increased use of medications, the composition and amount of good gut bacteria decrease even faster.
An optimum gut microbiota consists of a healthy balance of both good and bad gut bacteria species, which leads to good gut health.
Smoking is the biggest risk factor for COPD. Approximately 90% of COPD cases are caused by smoking [5]. Other risk factors for COPD include:
- Air pollution exposure - Secondhand smoke - History of childhood respiratory infections - Diet: dietary fiber intake is inversely related to COPD incidence [6]
A study published in the medical journal Gut in 2022 described the role of gut microbiota in COPD [7]. The study highlighted that dysbiosis (alteration of the gut microbiota) plays a role in influencing the pathogenesis of COPD. An increased abundance of Lachnospiraceae species in COPD patients may have contributed to chronic inflammation and worsening COPD. An increase in Lachnospiraceae species has also been observed in other chronic inflammatory diseases such as inflammatory bowel disease (IBD) [7].
The lung microbiota is known to play key roles in COPD [8]. Gut dysbiosis leads to increased gut permeability, leaking toxins and bacteria into the bloodstream which travel to the lungs, causing lung dysbiosis and possibly worsening COPD [8]. Increased gut permeability also leads to chronic systemic inflammation which worsens COPD.
For prebiotics which are dietary fibers, they also support healthy lung functions. By modifying our diets and increasing our intake of prebiotic fiber, we can potentially decrease our risk of COPD.
Prebiotics are food for our good gut bacteria. By supplementing our diets with prebiotics, we allow good gut bacteria to grow, and in turn crowd out bad ones. This establishes a healthy and balanced gut microbiota which helps support our respiratory health.
Good gut bacteria feeding on prebiotics also produce metabolites such as short-chain fatty acids (SCFAs) that confer health benefits. SCFAs are known to have anti-inflammatory effects and thus have the potential to reduce the risk of COPD [9].
The existence of the gut-lung axis suggests that improving our gut health supports our respiratory system and can potentially reduce the risk of lung diseases such as COPD. The use of prebiotics to induce positive changes to our gut microbiota and improve our gut health is one option we can consider to improve our respiratory health and reduce the risk of COPD.
 Introduction to the Gut-Lung Axis
Introduction to the Gut-Lung Axis
 Prebiotic: Gut Microbiota, Gut Health, and Beyond
Prebiotic: Gut Microbiota, Gut Health, and Beyond

- In recent years, researchers have begun to uncover the association between the gut microbiota and respiratory health via the gut-lung axis.
- Our lungs, which were traditionally thought to be sterile, are estimated to contain about 103 to 105 bacteria per gram of tissue.
- The gut and lungs communicate bi-directionally via their respective sets of microbiotas and the metabolites such as short-chain fatty acids (SCFAs) which are produced when good gut bacteria feeds on prebiotics.
Our lungs and respiratory system are vital for many bodily functions. Lung diseases remain as leading causes of death worldwide [1]. More people are now taking steps to maintain a healthy respiratory system and prevent lung diseases. In recent years, researchers have begun to uncover the association between the gut microbiota and respiratory health. Read on to find out more about the gut-lung axis.
Our gut consists of trillions of such microorganisms, including bacteria, viruses, fungi and protozoa species. This set of gut microbiota is unique to everyone and varies due to several factors including environmental, lifestyle, dietary habits, and consumption of medications such as antibiotics [2].
This innate set of gut microbiota (the set that we are born with) is thought to be the most optimum for oneself. This optimum composition deteriorates as we age. Along with poorer modern-day diets lacking in prebiotics (food for the good gut bacteria) as well as increased use of medications, the composition and amount of good gut bacteria decrease even faster.
An optimum gut microbiota consists of a healthy balance of both good and bad gut bacteria species, which leads to good gut health.
Recent technological advances have discovered the presence of microbes in our lungs, which were traditionally thought to be sterile [3]. In healthy lungs, it is estimated that there are about 103 to 105 bacteria per gram of tissue. This number is much lower compared to the lower gastrointestinal tract, where there are about 1011 bacteria cells per gram of tissue [3].
The colonization of bacterial species in the lungs depends on several factors such as air inhalation, migration of microbes from the oral cavity, and local conditions in the lungs such as pH, temperature and oxygen levels [3,4]. The bacterial species that reside in the lungs are similar to those in the gut, which consists of mainly Firmicutes and Bacteroidetes [5].
Although anatomically distinct, the gut and lungs communicate bi-directionally via their respective sets of microbiotas. This communication is referred to as the gut-lung axis [4]. Just as a healthy set of gut microbiota supports our cardiovascular health [6] and immune system [7], the health of the gut also affects our lungs.
Dysbiosis refers to an alteration of the gut microbiota and is associated with many inflammatory diseases both within and outside the gastrointestinal (GI) tract. Inflammatory diseases include inflammatory bowel disease (IBD) [8] and asthma [9]. Evidence of the gut-lung axis is further demonstrated in patients with chronic GI-related diseases such as IBD: these patients also tend to have a higher prevalence of respiratory-related diseases [10].
Other evidence that the gut and lungs communicate with each other [4]:
- Gut to lung microbiota: changes to an infant’s diet have been found to alter the lung microbiota - Lung to gut microbiota: influenza infection was found to induce changes in the gut microbiota, including a decrease in Lactobacillus species, a gut microbe species known to be beneficial to humans [11]
Good gut bacteria feed on prebiotics in a process called fermentation, which produces short-chain fatty acids (SCFAs). SCFAs such as butyrate and propionate have been found to have immunomodulatory and anti-inflammatory effects in the gastrointestinal tract [12].
These SCFAs also travel from the gut to the lungs, leading to local effects on the lungs such as modulating lung immune response and anti-inflammatory effects, which can potentially play a role in respiratory inflammatory diseases such as asthma [4]. Hence, dysbiosis at the gut level may affect immune responses in the lungs.
Ever since the COVID-19 pandemic started, more emphasis has been placed on the importance of lung health and ways to proactively support our lungs. The existence of the gut-lung axis shows that improving our gut microbiota through dietary changes and supplements such as prebiotics provides an opportunity to improve our lung health and protect against respiratory diseases such as asthma and COPD.
 Gut-Brain-Immune Axis: An Introduction
Gut-Brain-Immune Axis: An Introduction
 Prebiotic: Gut Microbiota, Gut Health, and Beyond
Prebiotic: Gut Microbiota, Gut Health, and Beyond
 Keeping Your Gut Microbiota Healthy to Combat Long COVID and Severe COVID-19 Infection
Keeping Your Gut Microbiota Healthy to Combat Long COVID and Severe COVID-19 Infection

- Recent studies have suggested the importance of our gut health and microbiota in lowering the risk of stroke.
- Alterations in gut microbiota compositions have been found in patients with stroke as compared to healthy individuals.
- Our gut health and microbiota also play important roles during post-stroke recovery by modulating several key parameters such as immune response, blood pressure, and blood sugar.
- The use of prebiotics to induce positive changes to our gut microbiota and improve our gut health is one option we can consider to reduce the risk factors for stroke.
Stroke is a disease that affects the cerebral arteries that supply blood to the brain [1]. It occurs when the blood and oxygen supply to the brain gets disrupted. This can happen due to a blood clot (ischemic stroke) or when the blood vessels rupture (hemorrhagic stroke) [1]. According to the U.S. Centers for Disease Control and Prevention (CDC), more than 795,000 people in the U.S. suffer from stroke every year [2].
Recent studies have suggested the importance of our gut health and microbiota in lowering the risk of stroke. World Stroke Day is held on 29 October every year to help raise awareness about this public health emergency [3]. Read on to learn more about how gut health and microbiota may affect stroke and post-stroke management, as well as how they can potentially be new modifiable risk factors for stroke.
Optimum gut health means having the right balance of good gut microbes and bad ones in our gut. This composition of microorganisms in our gut is collectively termed gut microbiota. Our gut consists of trillions of such microorganisms, including bacteria, viruses, fungi and protozoa species. This set of gut microbiota is unique to everyone at birth and changes during the course of your life due to several factors including environmental, lifestyle, dietary habits, and consumption of medications such as antibiotics [4].
The innate set of gut microbiota (the set that we are born with) is thought to be the most optimum for oneself. This optimum composition deteriorates as we age. Along with poorer modern-day diets lacking in prebiotics (i.e., food for the beneficial gut bacteria) as well as increased use of medications, the composition and amount of good gut bacteria decrease even faster.
Dysbiosis refers to an alteration of the gut microbiota. Poor gut health and dysbiosis have been found to negatively affect risk factors for stroke such as blood pressure [5], sugar [6] and cholesterol [7] levels. This can potentially lead to an increased risk of stroke.
Apart from mitigating risk factors for stroke, our gut health and microbiota may be directly related to stroke.
Dysbiosis is not only associated with poorer control of metabolic markers like blood pressure, sugar and cholesterol. Such alterations in gut microbiota compositions have also been found in patients with stroke as compared to healthy individuals. A study presented at the European Stroke Organisation Conference (ESOC 2022) identified certain types of bacteria such as Negativibacillus and Lentisphaeria that were associated with a more severe stroke in the acute phase [8].
Beneficial gut bacterial species feed on prebiotics in a fermentation process. This process produces metabolites such as short-chain fatty acids (SCFAs). SCFAs can regulate inflammation, blood pressure and sugar, which are all important risk factors for stroke. These metabolites have also been found to be in lower levels in patients with ischemic stroke as compared to healthy individuals [9].
Our gut health and microbiota also play important roles during post-stroke recovery by modulating several key parameters:
- Immune response: SCFAs produced from the fermentation of prebiotics can stimulate immune cells that are key factors in favorable stroke outcomes [10]. - Blood pressure: certain strains of beneficial gut bacteria have been identified to be associated with lowered blood pressure levels [11]. - Blood sugar: healthy gut microbiota and SCFAs can assist in blood sugar metabolism and homeostasis, including the cellular uptake of sugar and thus reducing blood sugar levels [6].
There is accumulating research showing the importance of gut health and microbiota in affecting the risk of stroke. Dysbiosis is associated with stroke and several risk factors of stroke such as blood pressure, sugar and cholesterol levels as well. The use of prebiotics to induce positive changes to our gut microbiota and improve our gut health is one option we can consider to reduce the risk factors for stroke.
This article is written in conjunction with World Stroke Day, which falls on 29 Oct every year.
 How Does Our Gut Health Affect Our Cardiovascular System?
How Does Our Gut Health Affect Our Cardiovascular System?
 Good Gut Health Is Linked To Lower Cholesterol Levels
Good Gut Health Is Linked To Lower Cholesterol Levels
 Gut Microbiota and Its Implications in Diabetes and Blood Sugar
Gut Microbiota and Its Implications in Diabetes and Blood Sugar

- Probiotics are defined by the International Scientific Association for Probiotics and Prebiotics (ISAPP) as live microorganisms that, when administered in adequate amounts, confer a health benefit on the host.
- Prebiotics are substrates that are selectively utilized by host microorganisms conferring a health benefit.
- Host microorganisms can refer to our innate gut microbiota and external probiotic supplements.
- Prebiotics help feed probiotic supplements, thereby increasing their chances of survival and proliferating successfully in the colon, preventing them from going to waste.
Probiotic supplements are widely used around the world. They are defined by the International Scientific Association for Probiotics and Prebiotics (ISAPP) as live microorganisms that, when administered in adequate amounts, confer a health benefit on the host [1]. In essence, probiotic supplements contain beneficial gut bacterial species, including Bifidobacteria and Lactobacillus species [2].
Probiotics are one of the members of the -biotics family. Other members include prebiotics, synbiotics and postbiotics. Read on to find out how another member of the -biotics family, prebiotics, helps feed your probiotic supplements and hence optimizes the effects of your probiotic supplements.
ISAPP has defined a prebiotic as a substrate that is selectively utilized by host microorganisms conferring a health benefit [3]. In the context of our gut, the host microorganisms may refer to:
- Our innate gut microbiota - External probiotic supplements
Prebiotics such as resistant starches and various types of oligosaccharides resist digestion from our human body’s enzymes and travel to the colon largely unchanged.
Our gut microbiota in the colon and the probiotic supplements we take will then feed on and ferment the prebiotics. This fermentation process produces metabolites such as short-chain fatty acids (SCFAs). SCFAs like butyrate confer health benefits such as providing a main source of energy for our colon cells and having anti-inflammatory properties [4].
This innate set of gut microbiota (the set that we are born with) is thought to be the most optimum for oneself. This optimum composition deteriorates naturally as we age. Along with poorer modern-day diets lacking in prebiotics (such as dietary fiber) as well as increased use of medications such as antibiotics, the composition and amount of good gut bacteria decrease even faster.
Having a set of healthy gut microbiota is important as it enhances and optimizes our gut health. Research has shown that our gut microbiota and health also affect systems outside of the gut. Our gut microbiota communicates with our brain bi-directionally via the gut-brain axis [5]. Good gut health also supports metabolic markers such as blood pressure [6], sugar [7] and cholesterol levels [8]. Gut microbiota and health also aid in supporting our cardiovascular [9], immune [10] and respiratory health [11].
The importance of gut health is increasingly being appreciated by people around the globe, as reflected in the increasing popularity of probiotic and prebiotic supplements.
Taking prebiotics helps to provide fuel for your innate gut microbiota. In addition, prebiotics also help feed the probiotic supplements, thereby increasing their chances of survival and proliferating successfully in the colon [12]. This will help prevent the probiotic supplements from dying and going to waste.
ISAPP recommends taking 3-5g of prebiotics a day to obtain the beneficial effects of prebiotics. Prebiotics can be obtained from foods (such as whole grains and legumes) or in the form of dietary supplements.
Probiotic supplements are widely used around the world. Increasingly, prebiotics are also taken together with probiotic supplements to increase their survivability and proliferation in the colon. This optimizes the effects of probiotic supplements and prevents them from going to waste. Taking prebiotics with probiotics will further enhance the health of our gut microbiota, thereby optimizing our overall gut health.
 Prebiotic: Gut Microbiota, Gut Health, and Beyond
Prebiotic: Gut Microbiota, Gut Health, and Beyond
 How Does Our Gut Health Affect Our Cardiovascular System?
How Does Our Gut Health Affect Our Cardiovascular System?

- Over 55 million people around the world are estimated to be living with dementia, with Alzheimer's Disease (AD) being the most common type.
- Although the exact causes of AD have yet to be fully established, amyloid and tau proteins are found to accumulate in the brain of patients with AD. Neuroinflammation is also often a key element in AD.
- Health of our gut microbiota is associated with AD. Harmful bacteria may worsen neuroinflammation, while metabolites from beneficial gut bacteria may help protect against it.
- Prebiotics could be used to enhance and regulate the health of the gut microbiota, thereby lowering the risk of developing AD.
Over 55 million people around the world are estimated to be living with dementia in 2020. This number is likely to double every 20 years, reaching a total of 78 million by the year 2030 [1]. The most common type of dementia is Alzheimer’s Disease (AD). We have discussed how dietary fiber may have effects on dementia based on a Japanese study done recently which found that a high intake of dietary soluble fiber is linked to a lower risk of developing disabling dementia [2]. Most prebiotics are dietary fibers. However, not all dietary fibers are prebiotics. Read on to find out more about the effects of prebiotics and gut microbiota on dementia and Alzheimer’s disease.
Our gut consists of trillions of such microorganisms including bacteria, viruses, fungi and protozoa species. This composition of microorganisms residing in our gut is collectively termed gut microbiota, which is unique to everyone. It varies due to several factors, such as environmental, lifestyle, dietary habits, and consumption of medications such as antibiotics [3].
This innate set of gut microbiota (the set that we are born with) is thought to be the most optimum for oneself. This optimum composition deteriorates as we age. Along with poorer modern-day diets lacking in prebiotics (i.e., food for the beneficial gut bacteria) as well as increased use of medications, the composition and amount of good gut bacteria decrease even faster.
Prebiotics are defined by the International Scientific Association for Probiotics and Prebiotics (ISAPP) as a substrate that is selectively utilized by host microorganisms conferring a health benefit [4]. In loose terms, prebiotics are food for the beneficial gut bacteria in a process called fermentation. This feeding process produces metabolites such as short-chain fatty acids (SCFAs) which confer health benefits to us. One such health benefit could be healthy aging.
Although the exact causes of AD have yet to be fully established, it has been found that various types of proteins (e.g., amyloid and tau proteins) accumulate in the brains of patients with AD [5]. Neuroinflammation is also a key element in the accumulation of amyloid hence the progression of AD [6].
The health of our gut microbiota is associated with AD. The gut microbiota and the brain communicate bi-directionally, and this interaction has been termed the gut-brain axis [7]. Dysbiosis, which refers to an altered gut microbiota, has been associated with various neurodegenerative diseases including AD. Reduction in microbial diversity has been observed in patients with AD as compared to patients without [8].
Harmful gut bacteria may even produce amyloid peptides, which can enter the bloodstream due to increased gut permeability. These peptides can then cross the blood-brain barrier and accumulate amyloid in the brain, further exacerbating AD [9]. Metabolites produced from harmful bacteria can also worsen neuroinflammation in the brain, while metabolites (such as SCFAs) produced from beneficial gut bacteria may help protect against AD by inhibiting the aggregation of amyloid peptides and maintaining cognitive functions [9,10].
Prebiotics are food selectively utilized by beneficial gut bacteria. By taking prebiotics, we improve the health of our gut microbiota by allowing the beneficial bacteria to grow and proliferate, at the same time crowding out the harmful ones. This may help to ameliorate the risk factors of AD such as gut permeability, amyloid peptides, and systemic inflammation [9].
There is currently no cure for AD. Current evidence points to the importance of the gut microbiota as a modulator of neurodegenerative diseases such as AD. The gut microbiota serves as a promising therapeutic target for the prevention and treatment of AD. Prebiotics could potentially be used to enhance and regulate the health of the gut microbiota, thereby lowering the risks of neurodegenerative diseases.
 Dietary Fiber and Its Links To Dementia
Dietary Fiber and Its Links To Dementia
 Prebiotic: Gut Microbiota, Gut Health, and Beyond
Prebiotic: Gut Microbiota, Gut Health, and Beyond

- Cardiovascular diseases are the leading causes of death worldwide, taking almost 18 million lives every year
- Optimum gut health means having a right balance of good gut microbes against bad ones in our gut.
- Our gut health is associated with our cardiovascular health in 3 ways.
- Prebiotics could be used to enhance and regulate the health of the gut microbiota, thereby improving gut health and support our cardiovascular health.
Cardiovascular diseases are the leading causes of death worldwide, taking almost 18 million lives every year [1]. This group of diseases is comprised of conditions affecting the heart and blood vessels, including coronary heart disease, heart attack, and stroke being the more common ones. Some of the risk factors for developing heart diseases are [2]:
- High blood pressure (hypertension) - High blood cholesterol - Diabetes - Obesity - Lack of physical activity - Excessive alcohol - Smoking
Lifestyle changes such as diet modifications and increasing physical activities are ways to lower our risks of developing heart diseases. Here we explore how our gut health and microbiota affect our cardiovascular system.
Optimum gut health means having a right balance of good gut microbes against bad ones in our gut. This composition of microorganisms in our gut is collectively termed as gut microbiota. Our gut consists of trillions of such microorganisms, including bacteria, virus, fungi and protozoa species. This set of gut microbiota is unique to everyone and varies due to several factors including environmental, lifestyle, dietary habits, and consumption of medications such as antibiotics [3].
The good gut bacteria feeds on prebiotics, leading to the production of metabolites such as short-chain fatty acids (SCFAs) which confer health benefits to us. These benefits include healthy blood sugar [4] and pressure levels [5], and immune health [6]. The growth of good gut bacteria will in turn crowd out the bad ones, leading to a healthy balance of gut microbiota and optimum gut health.
This innate set of gut microbiota (the set that we are born with) is thought to be the most optimum for oneself, and this composition and amount of good gut bacteria decrease as we age, along with modern-day diets lacking in prebiotics (food for the good gut bacteria) and increased consumption of medications such as antibiotics.
Although anatomically distinct, our gut is intrinsically linked to our heart. Dysbiosis, which refers to the alteration of the gut microbiota balance, has been reported in patients with risk factors for cardiovascular diseases, such as hypertension [7]. Disruptions in gut microbiota can also lead to excessive accumulation of body fat in early ages [8].
Here are 3 ways the health of our gut affects cardiovascular health and the various cardiovascular diseases [9,10].
#1: Production of TMAO and its adverse effects on cardiovascular health
Trimethylamine N-oxide (TMAO) has been found to be associated with an elevated risk of cardiovascular diseases, as shown by research done by Wang et al. in 2011, published in the Nature journal [11]. Elevated TMAO levels were shown to accelerate atherosclerosis, which is a significant pathway in the development of cardiovascular diseases [11]. Other studies across the US and Europe have also generated similar results with regards to TMAO and its potential to increase cardiovascular disease risks [12,13]. Other than atherosclerosis, TMAO is also a strong predictor of clinical outcomes in patients with heart failure [14].
TMAO is produced mainly via a multistep pathway with the gut microbiota as the starting point. Our gut microbiota metabolizes certain types of nutrients including choline, phosphatidylcholine and carnitine into a compound called trimethylamine (TMA). This process is carried out by an enzyme called TMA lyase, which is encoded by certain type of gut microbial genes. TMA is subsequently oxidized into TMAO by certain types of liver enzymes. The precursors of TMAO (choline, phosphatidylcholine and carnitine) are mainly found in red meats, egg yolk and other meat products. Altered gut microbiota can result in an increase in the production of such toxic metabolites such as TMAO.
#2: Leaky gut hypothesis
In a healthy and normal state, there are mechanisms to ensure intestinal barrier function, including tight junctions, mucus production and immunity [9]. In heart failure patients, there is often impaired intestinal barrier function due to bowel wall edema, leading to this ‘leaky gut’ hypothesis [9]. This leaky gut will lead to gut bacteria and other bacterial products into the bloodstream, which can result in pro-inflammatory states for the body. This is usually correlated with heart failure symptom severity and poorer outcomes [15]. Studies have also shown that heart failure patients have higher endotoxin levels in the bloodstream [16]. Such leaky gut can be improved by improving the health of your gut microbiota, which can be done via taking prebiotics [17].
#3: Effects of SCFAs
SCFAs are produced from the fermentation of prebiotics by the gut microbiota. These metabolites include butyrate, propionate, and acetate. SCFAs have been shown to modulate several risk factors involve in cardiovascular diseases, such as blood pressure [18], lipid metabolism [19] and blood sugar homeostasis [20]. By modulating and reducing these blood markers, risk of developing cardiovascular diseases can be reduced. Recent studies have also suggested that SCFAs directly influence cardiac activities, such as repair of heart tissues after injury (such as heart attacks) [21].
One way to improve our gut health and gut microbiota composition is to take more prebiotics. Prebiotics act as food for the beneficial gut bacteria allowing them to grow and crowd out the harmful ones. By optimizing the balance of our gut microbiota, we improve our gut health which in turn support our cardiovascular system, through less production of toxic metabolites such as TMAO and increase production of beneficial ones such as SCFAs.
Restricting the types of food that give rise to TMAO may also be another lifestyle intervention that can help reduce risk of cardiovascular disease. Reducing intake of red meat can reduce our dietary carnitine and choline, thereby reducing TMAO levels as well.
Other than the benefits on cardiovascular health, optimum gut health can also support the health of other body functions and systems, such as:
- Metabolic markers: blood pressure, sugar and cholesterol - Immune health - Nervous system - Mental health - Healthy aging - Respiratory health - Skin health
Cardiovascular diseases such as coronary heart disease, heart attacks and stroke are some of the most common diseases and causes of death worldwide, and cost billions of dollars in healthcare expenditure. Besides reducing your risks through lifestyle modifications, you can also consider optimizing your gut health. Taking prebiotics enhances the beneficial gut microbiota, which in turn helps improve gut health leading to reduction in risks of cardiovascular diseases.
 Gut Health and Blood Pressure (Hypertension): How Are They Related?
Gut Health and Blood Pressure (Hypertension): How Are They Related?
 Prebiotic: Gut Microbiota, Gut Health, and Beyond
Prebiotic: Gut Microbiota, Gut Health, and Beyond
 Good Gut Health Is Linked To Lower Cholesterol Levels
Good Gut Health Is Linked To Lower Cholesterol Levels